|
Secure PDF Files Secure PDF files include digital rights management (DRM) software. DRM is included at the request of the publisher, as it helps them protect their copyright by restricting file sharing. In order to read a Secure PDF, you will need to on your computer. GAMP 5 - Good Automated Manufacturing Practice. How the White Paper about GAMP 5 can help You. Lundi, 19 juin 2017 Gamp5 Guidelines Pdf GAMP 5 - Good Automated Manufacturing Practice. How the White Paper about GAMP 5 can help You. The International Society for Pharmaceutical Engineering (ISPE) issued the worldwide. • PIC/S Good Practice for Computerised Systems in regulated “GXP” Environments www.picscheme.org • Eudralex Volume 4 GMP Guide Annex 11: Computerised Systems • GAMP 5 - Good Automated Manufacturing Practice 28th September 2012. The FileOpen Plug-In works with Adobe Reader and other viewers. What you can do with a Secure PDF:• Print • Search • Highlight • Bookmark Please note that some publishers - including BOMA, IADC and NRC - do not allow printing of their documents. • • Immediate download • $570.00 •. Please note: This is the English version of GAMP 5. A version of this publication in German GAMP® 5 provides pragmatic and practical industry guidance to achieve compliant computerized systems fit for intended use in an efficient and effective manner. This technical document describes a flexible risk-based approach to compliant GxP regulated computerized systems, based on scalable specification and verification. It points to the future of computer systems compliance by centering on principles behind major industry developments such as PQLI; ICH Q8, Q9, Q10;. This revolutionary Guide addresses the entire lifecycle of an automated system and its applicability to a wide range of information systems, lab equipment, integrated manufacturing systems, and IT infrastructures. Buckwild presents ep rar. It contains new information on outsourcing, electronic batch recording, end user applications (such as spreadsheets and small database applications), and patch management. An exciting new innovation is the CD accompanying the softcover Guide. It provides supporting materials, including differences between GAMP® 4 and GAMP® 5, key diagrams, templates, forms, example documents, and background information. (CD is Windows compatible only.). If you order the secure PDF, these supplemental materials are included as downloads. Product Details Edition: 5th Published: ISBN(s): Number of Pages: 356 File Size: 1 file, 4.2 MB Browse related products from International Society for Pharmaceutical Engineering •. 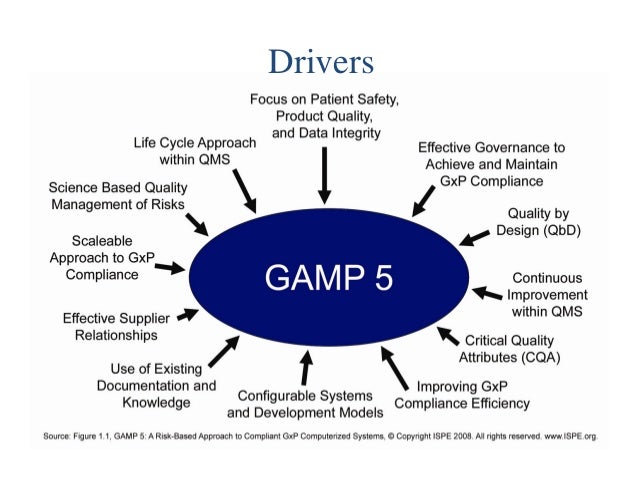
GAMP 5 - Good Automated Manufacturing Practice MasterControl GAMP 5 Offers Good Automated Manufacturing Practice Guidelines for Manufacturing Companies to Comply with Regulated Environments MasterControl's Quality Management Systems are designed by industry practitioner for automating the GAMP 5 process in any organization. A Quality Management Software system is the crux of any quality and compliance process. It is a regulatory requirement that FDA / global regulatory inspectors and ISO auditors consider critical. An automated GAMP 5 system reduces audit time and findings, and a decreases risk of product recalls. It improves product quality and safety, increases customer satisfaction, and ensures FDA and ISO compliance. How the White Paper about GAMP 5 can help You The International Society for Pharmaceutical Engineering (ISPE) issued the worldwide release of GAMP 5: A Risk-Based Approach to Compliant GxP Computerized Systems in March of 2008. The updated manual for GAMP 5 provides guidelines for good automated manufacturing practices that are referenced in markets around the world, including North and South America, Europe, and Japan.
0 Comments
Leave a Reply. |
- Blog
- Home
- Om shanti oshana movie
- Autocad student
- Brew cask freetype
- Building estimate pdf file
- Startup disk creator download
- Java ver 7 update 45
- Kamen rider ooo henshin pose
- Basilisk anime nudity
- Karthik tamil actor movie aruvian
- Mac os 7-5 3
- How to register fl studio alpha mac
- Rolando rodriguez nato
 RSS Feed
RSS Feed
